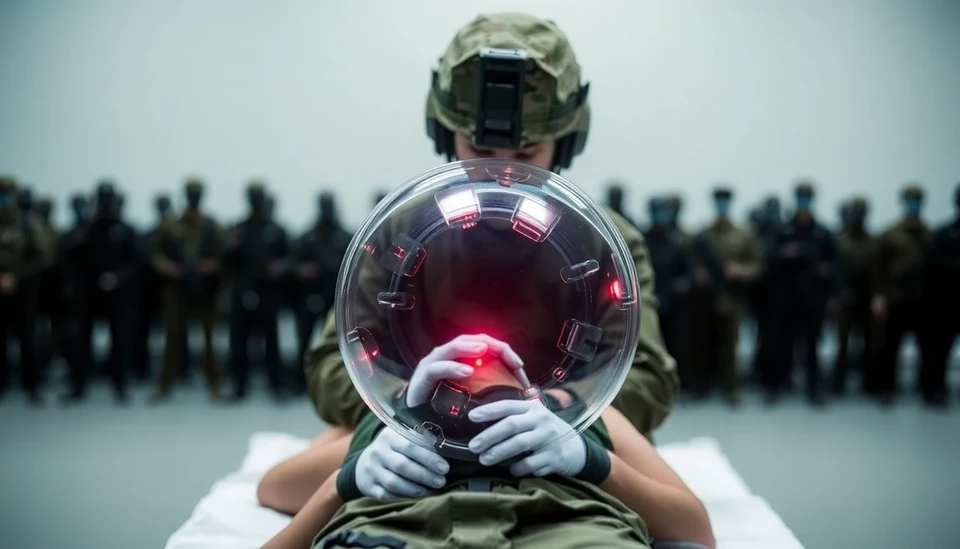
US Approves Controversial Device for Battlefield Injuries Amid Safety Concerns
The United States has recently given the green light to a new medical device aimed at treating battlefield injuries, a decision that has raised significant concerns among health experts and former regulatory officials. The approval, granted by the Food and Drug Administration (FDA), comes despite warnings about the potential serious risks associated with the device.
Continue reading
Medical Device Industry Rallies Against FDA Worker Firings Amidst Unprecedented Challenges
The medical device sector has recently erupted in a wave of protests following the controversial dismissal of several key employees within the U.S. Food and Drug Administration (FDA). This situation has ignited debates over the regulatory body’s capabilities and the broader implications for public health and innovation in the industry.
Continue reading
FDA Officer Overseeing Musk's Neuralink Devices Terminated Amid Controversy
In a surprising turn of events, the U.S. Food and Drug Administration (FDA) has terminated an official who played a crucial role in overseeing Neuralink, the neurotechnology company founded by Elon Musk. The dismissal has raised eyebrows and concerns about the ongoing regulatory scrutiny that Neuralink faces as it aims to develop cutting-edge brain-computer interface devices.
Continue reading
Kestra Files for IPO Amidst Market Buzz and Future Aspirations
Kestra, a leading medical device company backed by Bain Capital and Endeavour, has officially filed for an initial public offering (IPO). This move comes at a significant time in the healthcare sector, as the demand for innovative medical technologies continues to surge. The company has positioned itself as a strong player in the industry, focusing on developing advanced medical solutions that cater to a wide range of healthcare needs.
Continue reading
Abbott's 2025 Profit Forecast Aligns with Market Expectations, Driven by Surge in Device Demand
Abbott Laboratories has recently announced a profit forecast for 2025 that is in line with market expectations, primarily driven by a surge in demand for its medical devices. The company, known for its extensive range of healthcare products, revealed that its strong outlook is bolstered by recovery in hospital settings and an increase in outpatient procedures.
Continue reading
EU Investigation Uncovers Discriminatory Practices in Chinese Medical Device Procurement
In a recent report released by the European Union, evidence has emerged indicating that Chinese authorities are systematically discriminating against foreign suppliers in the procurement of medical devices. This investigation highlights a growing concern among European nations regarding unequal trade practices and market access issues in China, particularly in the healthcare sector, which is critical in the context of global health and economic recovery.
Continue reading
Stryker to Acquire Inari Medical in Major $80 per Share Deal
In a significant development within the medical device sector, Stryker Corporation has announced its agreement to acquire Inari Medical for $80 per share, marking a notable step forward in the expansion of its vascular portfolio. The acquisition deal, valued at approximately $4 billion, aims to bolster Stryker’s offerings in the treatment of venous diseases, which is a rapidly growing area of healthcare.
Continue reading
Abbott Adjusts 2024 Projections, Fuelled by Medical Device Innovation
Abbott Laboratories recently updated its financial forecasts for the year 2024, indicating a narrowed range that reflects its ongoing commitment to expanding its medical devices segment. The company's revised outlook suggests robust growth driven by an increased demand for its innovative healthcare solutions, particularly in the diagnostic and therapeutic sectors.
Continue reading